Advancing Safe Elimination Strategies for Onchocerciasis in Loa loa Co‑Endemic Areas












Background
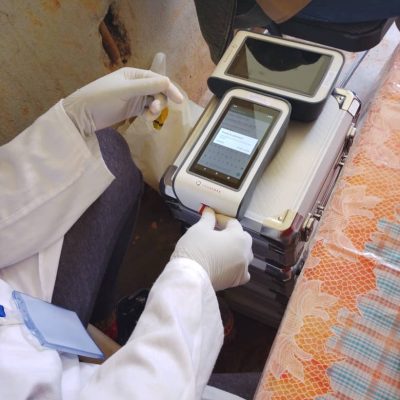
Elimination of onchocerciasis in hypo‑endemic areas co‑endemic with Loa loa remains a major challenge. The risk of serious adverse events (SAEs) when ivermectin (IVM) is administered to individuals with high Loa loa microfilarial loads (>30,000 mf/mL) has prevented WHO from recommending mass drug administration (MDA) in these settings. The LoaScope, a mobile, point‑of‑care diagnostic first validated in 2015, enables rapid quantification of L. loa infection intensity and supports a ‘Test‑and‑Not‑Treat’ (TaNT) strategy. Earlier generations of the LoaScope were successfully deployed in Cameroon, Nigeria, and Gabon. A third‑generation (G3) LoaScope has now been developed to improve durability, data processing, and field usability, and to enable scale‑up of TaNT.
Objective
To validate the field performance of the third‑generation LoaScope against conventional calibrated thick blood smear (CTBS) prior to large‑scale research and programmatic use, including assessment of diagnostic accuracy, reliability (intra‑/inter‑device and operator variability), and operational usability for TaNT.
Methods
A cross‑sectional survey was conducted in the Awae Health District (Cameroon) between September–October 2023. Volunteers aged ≥10 years underwent interviews, parasitological testing (CTBS and LoaScope), and clinical assessment (RAPLOA). The performance G3 LoaScope was evaluated against CTBS as the gold standard. Inter‑ and intra‑device reliability, operator variability, and user feedback were also assessed.
Results
A total of 1,146 individuals (median age 43 years) were enrolled. The prevalence of loiasis was 30.9% by CTBS and 23.2% by LoaScope. Infection intensities were broadly comparable, though LoaScope consistently reported lower mf densities. Agreement between LoaScope and CTBS was moderate to substantial (Kappa ~0.66–0.72), with sensitivity around 66% and specificity near 99%. While LoaScope reliably detected infections, it identified fewer very high‑intensity cases (≥20,000 mf/mL) than CTBS (16 vs. 40). Reliability testing showed substantial intra‑device and almost perfect inter‑device agreement. User feedback was highly positive, though minor technical issues (screen freezing, capillary ejection, slow synchronization) were noted.
Concluding Remarks
The G3 LoaScope demonstrates strong reliability and specificity, with substantial ability to detect L. loa infections at point of care. While its discrimination of very high microfilarial densities remains lower than CTBS, the device is a critical tool for safely expanding ivermectin MDA in hypo‑endemic areas. Combined with statistical modeling and antibody diagnostics, the LoaScope offers a pathway toward a WHO‑endorsed strategy for onchocerciasis elimination in Loa loa co‑endemic regions. Ongoing refinements to address technical dysfunctions will further enhance its programmatic utility.
Our Location
- 285, Street 1.411, Fouda Quarter, Yaounde 5, Cameroon P.O. Box 5797 Yaounde
- +237 6 75 06 91 58
- stephanie.jouego@ismcm.org
Copyright © 2026 Made by ISM Cameroon